 |
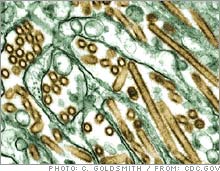
| Colorized transmission electron micrograph of Avian influenza A H5N1 viruses (seen in gold) grown in MDCK cells (seen in green). |
|
|
|
NEW YORK (CNN/Money) -
The U.S. government is getting set to add billions of dollars to its budget to build a stockpile of drugs to fight the threat of a deadly avian flu virus, and European drugmakers are considered the top candidates for federal funds.
But White House officials will meet with representatives from the U.S. pharmaceutical industry Friday to encourage them to get involved in making flu vaccine amid fears of an avian flu pandemic, CNN has learned.
Most U.S. drugmakers have stopped making flu vaccines for a variety of reasons, but many public health advocates believe having a reliable supply of the vaccine may be the best way to contain a "bird flu" pandemic in humans.
The avian flu, also known as H5N1, has killed more than 60 people in Asia. Some public health experts are concerned that it could surface here, particularly after scientists announced Wednesday they had reconstructed the influenza virus that killed up to 50 million people in 1918 -- and discovered that it was a bird flu that had jumped to humans.
The Senate has approved nearly $4 billion to stockpile medications to protect the populace against avian flu, said Jim Manley, spokesman for Senate Minority Leader Harry Reid, the Nevada Democrat who's one of the lead sponsors for the budget amendment.
The amendment, which needs to be reconciled with a similar measure passed by the House, is in addition to the $5.6 billion Bioshield budget that's being used to stockpile drugs in preparation for the threat of terrorist attack.
The funding would be added to the Department of Defense budget "to stockpile anti-virals and necessary medical supplies" and "expand global surveillance and domestic vaccine infrastructure," according to Sen. Reid's Oct. 4 letter to President Bush seeking his support.
The additional money would also be used to improve emergency preparedness centers and hospitals, as well as "risk communication" to the American public.
Roche is top candidate
Roche Holding AG (up $2.11 to $144.03, Research), the Swiss maker of Tamiflu, is considered a top candidate for a government contract because its anti-viral drug is already on the market, analysts said.
"If they want to stockpile something that works, Tamiflu is the way to go, because the data is better [than its competitors] in being able to treat the bird flu," said Gbola Amusa, an analyst at Bernstein & Co.
Andreas Theisen, analyst for German bank WestLB, estimated that Roche will receive $1.7 billion to $1.8 billion from the U.S. government to produce Tamiflu, and Roche has ramped up its manufacturing capacity in the last two years in preparation for massive orders.
Tamiflu, an orally administered pill also known as oseltamivir, must be taken within two days of infection in order to be effective in people with avian flu.
British drug giant GlaxoSmithKline (up $0.46 to $51.41, Research) has a competing product, Relenza, an inhalable pharmaceutical also known as zanamivir.
Theisen said Tamiflu is easier to use than Relenza, making it the top choice for the feds. Also, Relenza can't be used by asthma sufferers, said Theisen, bolstering its rival's odds of getting the winning bid.
"The pill is the thing to go for," said Theisen, referring to Tamiflu.
Vaccines might be an even more important factor in the budget because they are used to prevent the onset of viruses before they sweep through a population, said Amusa, the Bernstein analyst.
Up to $3 billion
Amusa estimated that the U.S. government will spend $2.5 billion to $3 billion to stockpile vaccines to prevent avian flu, and French drug giant Sanofi-Aventis (up $0.13 to $42.03, Research) is the top candidate.
"To have an immunized population is always ideal," said Amusa. "Sanofi would clearly benefit. They produce about half the world's flu vaccine."
Unlike Roche, Sanofi-Aventis does not have an avian flu drug approved by the Food and Drug Administration. But this scarcely matters, as the government does not require the FDA stamp of approval to acquire drugs that fulfill a national need.
And the feds have already given Sanofi-Aventis a vote of confidence. On Sept. 15, the Department of Health and Human Services agreed to pay $100 million to Sanofi-Aventis to manufacture its vaccine at the company's factory in Swiftwater, Pa. This is the company's fifth avian flu contract with the U.S. government, though the previous contracts were considered "investigational."
Still, these drugmakers could easily get bogged down by large government contracts if they lack capacity to produce the drugs, the analysts said.
"People are really starting to get nervous," said Theisen, who has seen colleagues in Germany buy avian flu vaccines for their families. "If [avian flu] really materializes, it will be hard to get anything.
The analysts interviewed for this story do not own stock in the companies mentioned here.
To read about Operation Bioshield, click here.

|