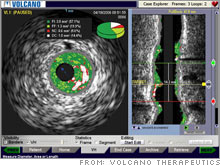
Medical companies take 'Fantastic Voyage' into heartTwo makers of high-tech heart screening seem to be getting a boost due to stents and blood clot risk.NEW YORK (CNNMoney.com) -- The trouble with drug-coated stents could be a boon to Volcano and other companies specializing in high-tech heart screening that could have been dreamed up in the sci-fi classic "Fantastic Voyage." Volcano (down $0.06 to $18.85, Charts), which went public last year, makes catheters that snake through arteries and take ultrasound pictures. But instead of Raquel Welch in a space suit, the Volcano cameras take color images of stents and cholesterol build-up. The catheter can be used to assess whether stents have been properly placed and to detect clogged arteries.
"We want to eliminate and solve heart attacks," said Scott Huennekens, chief executive of five-year-old Volcano, based in Rancho Cordova, Calif. Volcano sells its $65,000 devices as part of a $1 million catheter lab marketed by partner General Electric. Another partner, Johnson & Johnson (down $0.02 to $60.51, Charts), markets the device in Japan, an active market for Volcano. Boston Scientific (down $0.15 to $14.83, Charts), Phillips and Siemens make competing cath lab systems. Volcano's catheter-camera, technology known as intravascular ultrasound (IVUS), seems to be getting a lift from problems that hit the nation's $3 billion stent industry in 2006. Volcano launched its IPO in June, six months before the Food and Drug Administration held a safety meeting on the newest type of stents, which had been blamed for causing blood clots. "IVUS as a technology has been around for well over a decade, but there wasn't a real driver to use it," said Jose Haresco, analyst at Merriman, Curhan, Ford & Co., a financial services company in San Francisco. "Stenting seemed to be going well. Now, all of a sudden, stenting isn't going that well. Things are not as rosy as we thought" in the drug-coated stent business. Volcano, a small company with a market cap of about $700 million, saw sales grow 12 percent to $103 million last year. But sales jumped 20 percent in the fourth quarter. The increase came in the same quarter that advisers to the FDA held a two-day meeting on drug-coated stents. The experts said that while people with drug-coated stents are at higher risk for blood clots, the cause was unclear. But they also called for a warning about blood clot risks when doctors insert the stents in cases where it's not approved by the FDA. More than half of drug-coated stent procedures are in such "off-label" uses. Stents are metal tubes that doctors insert into a patient's arteries after a heart attack to unclog the arteries and keep the blood flowing normally. Stents are also used in some high-risk patients to prevent heart attacks and it's common for patients to have as many as four or five stents in their bodies at once. Boston Scientific, Medtronic (Charts), Johnson & Johnson andAbbott Labs (Charts) compete in that business. The first generation of stents, made of bare metal, are still widely used. Then, in 2003, the FDA approved drug-eluding stents, which are coated with anti-inflammatory drugs to lessen abrasiveness against the artery walls. But in an unexpected blow to the industry, the drug-coated stents have been blamed for causing blood clots, which they're supposed to prevent. No one seems to know why this happens, or why bare metal stents don't have this problem. Haresco of Merriman, Curhan, Ford & Co. said the trouble with stents might have boosted sales for Volcano: "There's more demand for the [IVUS] product and more demand to monitor the patient: to accurately look at a patient before you put a stent in, while you put a stent in, and after you put a stent in," said Haresco. Volcano's color-coded system of artery monitoring - clogging shows up in ultrasound imaging as tell-tale red - can be used to detect impending blood clots in high-risk patients. Monitoring the status of patients at high-risk for heart attacks is a potentially lucrative market. Heart disease is the No. 1 killer in America and the baby boomer-fueled industry for heart treatment is estimated at $85 billion in the United States, including medication, products like stents, surgery and other treatments. Meanwhile, another company called Acusphere (up $0.01 to $2.79, Charts) is working on a different system to monitor heart disease risk called Imagify. Imagify uses microscopic gas-filled spheres that are injected into the blood, instead of traditional dyes, to amplify the effects of ultrasound used to detect clogged arteries. Acusphere CFO John Thero said his company is conducting late-stage tests for its microsphere technology and plans to file for FDA approval later this year. Thero said he wants Imagify technology to be used in conjunction with ultrasound equipment made by Phillips, which accounts for about a quarter of the $2.4 billion market for high-tech cardiovascular screening, and to compete with radiation screening technology made by GE, which controls the rest. Unlike Volcano, Acusphere doesn't have its device on the market because it's still in testing, and investing in this company is betting that its study results will be strong. "This is a high-risk stock because the product does need to report good study data," said Angela Maria Larson, analyst for Susquehanna Financial Group. She expects test results to be announced in the second quarter. Acusphere, based in Watertown, Mass., faces considerable risks involving the success of Imagify, analysts said, noting that a company with a similar microsphere technology, closely held Point Biomedical Systems, failed to satisfy the FDA due to unimpressive late-stage test results. Larson and Ian Sanderson, analyst for Cowen & Co., both project annual Imagify sales of about $200 million by 2010. But Sanderson said Acusphere is not revolutionary and will have a tough time penetrating the market for so-called nuclear heart screening, which uses radioactive tracers to track blood flow. "[Imagify] makes an old technology a little better," said Sanderson, who projects Acusphere will take no more than 3 percent of market share away from nuclear coronary scans, or 300,000 of the 9 million annual procedures. "We're not assuming tremendous penetration." The analysts quoted in this story do not own shares of stocks mentioned here, but Cowen has conducted banking for Acusphere and Merriman Curhan Ford makes a market in Volcano. |
Sponsors
|