 Tylenol maker McNeil-PPC is hit with fourth recall in seven months.
Tylenol maker McNeil-PPC is hit with fourth recall in seven months.
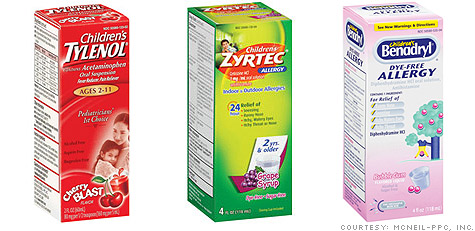
NEW YORK (CNNMoney.com) -- The maker of several over-the-counter drugs, including Tylenol, Motrin and Benadryl, said Monday that it is taking "corrective actions" at its U.S. plant after some 40 children's versions of those drugs were recalled for quality concerns.
McNeil-PPC, the division of Johnson & Johnson (JNJ, Fortune 500) that manufactured the recalled products, has been hit with a string of major recalls. The latest one is its fourth in the past seven months.
McNeil said on Saturday that it took action as a "precautionary measure," saying that some of the products could have problems with their ingredients while others could contain "tiny particles."
On Monday, McNeil elaborated on the nature of the "tiny particles," saying they may be solidified product ingredients or manufacturing residue such as tiny metal parts.
"We're conducting a comprehensive quality assessment across our manufacturing operations," said Johnson & Johnson spokesman Marc Boston. "Corrective actions will be implemented before new manufacturing is initiated at our Fort Washington, Pennsylvania plant where the recalled products were made."
McNeil's Pennsylvania plant is the only plant that manufactures Consumer Healthcare liquid pediatric products.
In November 2009, the company recalled five lots of its Tylenol Arthritis Pain 100 count with the EZ-open cap product due to reports of an unusual moldy, musty, or mildew-like odor that led to some cases of nausea, stomach pain, vomiting and diarrhea.
In December, McNeil expanded that recall to include all available product lots of Tylenol Arthritis Pain caplet 100 count bottles with the red EZ-open cap.
In January, McNeil recalled an undisclosed number of containers of Tylenol, Motrin and other over-the-counter drugs after consumers complained of feeling sick from an "unusual" odor.
The drugmaker said the smell was caused by a chemical called "2,4,6-tribromoanisole (TBA)," which is applied to wooden pallets that are used to transport and store packaging materials. But the U.S. wooden pallet industry took issue with McNeil's explanation.
More details about what's wrong with the drugs would be made available once the the Food and Drug Administration (FDA) has reviewed its inspection of the plant, the agency said.
"The actual inspection finished on Friday and we're hoping to get the complete report of our inspection this week," said Elaine Ganz Bobo, a spokeswoman with the FDA.
She said the agency decided to push for the recall before releasing all the details of the inspection because of the "vulnerability" of the consumers of these drugs.
"These are over-the-counter drugs and they impact infants and children," she said. "So it was even more important to get the message out that there could be a potential problem."
As of Monday, she said the FDA still maintains its position that the recalled drugs pose a "remote" potential for serious health problems.
Meanwhile, Boston said consumers could either email or call the company to get a refund or coupon for the value of the product. ![]()






| Index | Last | Change | % Change |
|---|---|---|---|
| Dow | 32,627.97 | -234.33 | -0.71% |
| Nasdaq | 13,215.24 | 99.07 | 0.76% |
| S&P 500 | 3,913.10 | -2.36 | -0.06% |
| Treasuries | 1.73 | 0.00 | 0.12% |
| Company | Price | Change | % Change |
|---|---|---|---|
| Ford Motor Co | 8.29 | 0.05 | 0.61% |
| Advanced Micro Devic... | 54.59 | 0.70 | 1.30% |
| Cisco Systems Inc | 47.49 | -2.44 | -4.89% |
| General Electric Co | 13.00 | -0.16 | -1.22% |
| Kraft Heinz Co | 27.84 | -2.20 | -7.32% |
|
Bankrupt toy retailer tells bankruptcy court it is looking at possibly reviving the Toys 'R' Us and Babies 'R' Us brands. More |
Land O'Lakes CEO Beth Ford charts her career path, from her first job to becoming the first openly gay CEO at a Fortune 500 company in an interview with CNN's Boss Files. More |
Honda and General Motors are creating a new generation of fully autonomous vehicles. More |
In 1998, Ntsiki Biyela won a scholarship to study wine making. Now she's about to launch her own brand. More |
Whether you hedge inflation or look for a return that outpaces inflation, here's how to prepare. More |