Search News
 Drug recalls hit a new record in 2009, with one company accounting for more than 1,000 of them.
Drug recalls hit a new record in 2009, with one company accounting for more than 1,000 of them.
NEW YORK (CNNMoney.com) -- Recalls of prescription and over the counter drugs are surging, raising questions about the quality of drug manufacturing in the United States.
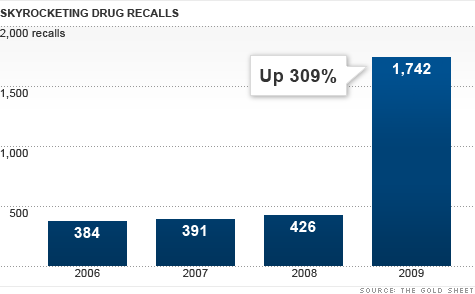
The Food and Drug Administration reported more than 1,742 recalls last year, skyrocketing from 426 in 2008, according to the Gold Sheet, a trade publication on drug quality that analyzes FDA data.
One company, drug repackager Advantage Dose, accounted for more than 1,000 of those recalls. Even excluding Advantage Dose, which has shut down, recalls jumped 50% last year.
"We've seen a trend where the last four years are among the top five for the most number of drug recalls since we began tallying recalls in 1988," said Bowman Cox, managing editor of the Gold Sheet. "That's a meaningful development."
The fast pace of drug recalls seems to be continuing in 2010. Drug recalls totaled 296 from January through June of this year, said Cox. "If we continue at this same rate, we could get 600 or more recalls by the end of the year," he said. "That's still a very high rate of recalls."
High-profile recalls of Tylenol and other products by McNeil Consumer Healthcare, a unit of Johnson & Johnson (JNJ, Fortune 500), have drawn attention to quality concerns in manufacturing.
The mounting problems over recalls have also drawn attention from lawmakers. Two bills introduced this year would impose stricter regulations on the industry and give the FDA authority to mandate recalls.
The spike in recalls, especially of generic and over-the-counter drugs, is being driven by manufacturing lapses, experts say. Some of the biggest culprits: the quality of raw materials, faulty labeling and packaging and contamination.
Increased FDA scrutiny of drug production facilities may also be triggering recalls.
For its part, the FDA maintains that it hasn't identified any alarming patterns in last year's recalls.
An FDA official told the Gold Sheet that 2009 saw no increase in recall "incidents" -- or recalls involving one product problem at a company -- just more recalls per incident.
"Since every recall situation is unique, it would be difficult to assess whether there are any trends or increases in recalls this year," FDA spokeswoman Elaine Gansz Bobo told CNNMoney. "At this time, however, we have not identified any trends."
A number of factors are fueling the recall surge. The stampede by drugmakers to be first to bring generic versions to market, after drugs lose patent protection, is one of them.
"The first applicant typically gets the lion's share of the business for the new drug," said Cox.
Generic drugs account for about three quarters of all prescription drug sales, according to industry group PhRMA.
In their hurry, drugmakers sometimes fail to spend enough time time learning how best to make the drug.
For example, Detroit-based Caraco Pharmaceutical Laboratories issued four "tablet-thickness" recalls on March 31, 2009, an additional one on June 8 and yet another recall on April 17 for products that might have been contaminated, said Cox.
"So they get the application. They make and market the drug, but they could still have problems down the road if they haven't really understood the optimum way to make that drug," said Cox.
Caraco could not be reached for comment.
Drugmakers aren't the only ones that have run into problems. Repackagers that relabel drugs into smaller units for resale or distribution to health care facilities, have also had serious lapses.
Advantage Dose, which led last year's recalls by far, is one such company. It issued 1,106 recalls last year following the FDA's inspection of its facility in late 2008. According to the Gold Sheet report, the FDA's inspection listed a wide range of manufacturing problems.
Another reason for the recalls: Cost cutting that goes too close to the bone.
Drugmakers, facing intense competition, are trimming manufacturing investment or outsourcing production, said Prabir Basu, executive director of the National Institute for Pharmaceutical Technology and Education, a nonprofit scientific and research organization.
"It is very expensive to make drugs. It also costs a lot of money to maintain adequate quality controls," Basu said. "You have to invest a lot."
And since generic and over the counter drugs aren't as lucrative for drugmakers as prescription drugs, companies may not be investing enough resources to make high-quality, safe products.
"We still have the best at drug manufacturing here in the United States. But if we're seeing signs of trouble, imagine what's happening outside of the U.S. in other countries from where we import drugs," said Basu.
The Gold Sheet report said that 165 recalls last year were of products made, or believed to be made, abroad. That was up 58% from 2008.
"Overall, I am concerned about the quality of drugs," said Basu. ![]()






| Index | Last | Change | % Change |
|---|---|---|---|
| Dow | 32,627.97 | -234.33 | -0.71% |
| Nasdaq | 13,215.24 | 99.07 | 0.76% |
| S&P 500 | 3,913.10 | -2.36 | -0.06% |
| Treasuries | 1.73 | 0.00 | 0.12% |
| Company | Price | Change | % Change |
|---|---|---|---|
| Ford Motor Co | 8.29 | 0.05 | 0.61% |
| Advanced Micro Devic... | 54.59 | 0.70 | 1.30% |
| Cisco Systems Inc | 47.49 | -2.44 | -4.89% |
| General Electric Co | 13.00 | -0.16 | -1.22% |
| Kraft Heinz Co | 27.84 | -2.20 | -7.32% |
|
Bankrupt toy retailer tells bankruptcy court it is looking at possibly reviving the Toys 'R' Us and Babies 'R' Us brands. More |
Land O'Lakes CEO Beth Ford charts her career path, from her first job to becoming the first openly gay CEO at a Fortune 500 company in an interview with CNN's Boss Files. More |
Honda and General Motors are creating a new generation of fully autonomous vehicles. More |
In 1998, Ntsiki Biyela won a scholarship to study wine making. Now she's about to launch her own brand. More |
Whether you hedge inflation or look for a return that outpaces inflation, here's how to prepare. More |